Biomarkers In Peripheral Biofluids
For the central nervous system
Difficulties in diagnosing neurologic diseases
Diagnosing neurologic diseases, monitoring their progression, and evaluating the efficacy of treatments for these diseases is notoriously difficult. In most cases, the diagnosis, evaluation of disease progression, and assessment of treatment effectiveness rely on clinical evaluation, which is error-prone because patient-to-patient variability is high and because the symptoms of many diseases overlap, especially at early stages. To improve the diagnostic accuracy, many labs search and develop biomarkers. However, obtaining reliable biomarkers is difficult due to the inaccessibility of the brain. Two approaches have been used traditionally – brain imaging and analysis of cerebrospinal fluid (CSF). However, both approaches suffer from important drawbacks. Brain imaging in many cases is not accurate enough and the associated costs are high. The more informative imaging techniques for soft tissues, such as MRI or PET scans, are done typically in large medical centers and therefore are less accessible in rural communities or small towns. CSF analysis requires a lumbar puncture, which is relatively invasive and refused by many patients, particularly when multiple tests are needed over time for monitoring disease progression or treatment effect.

The symptoms of many neurodegenerative diseases overlap, especially at early stages. Is it Parkinson's Disease (PD) or multiple system atrophy (MSA)? Or it is progressive supranuclear palsy (PSP) or corticobasal degeneration (CBD)? Symptoms also vary from patient to patient. Together, overlapping symptoms and patient variability complicate diagnosis, evaluation of disease progression, and assessment of treatment effectiveness.
Better biomarkers with more accessible body fluids
To address these issues, multiple groups have been testing ways to measure biomarkers in more accessible body fluids, such as blood, urine, or saliva, or in easily accessible organs, such as the skin or the eye. In many cases, substantial differences in levels of particular biomarkers have been observed on average between patients and healthy controls or among patients with closely related diseases. However, even when on average the groups differ significantly, there can be considerable overlap among them, resulting in low sensitivity and specificity of the test.
A promising approach is to analyze biomarkers in extracellular vesicles (EVs)/exosomes originating in the central nervous system (CNS) and isolated from the plasma or serum. This approach provides a window into changes in the biochemistry of the brain using a simple blood test. The methodology involves first isolation of all the EVs from the plasma/serum by chemical precipitation, followed by immunoprecipitation using antibodies against specific brain markers, which are present on the surface of the EVs originating in the CNS.
Using a simple blood test for our research
Our main research direction using this approach has been for distinguishing among parkinsonian disorders, which display a large symptom overlap in early stages. In particular, we found that by measuring α-synuclein concentration levels in EVs originating in both neurons and oligodendrocytes, and comparing the levels between the two, we could distinguish between Parkinson’s disease and multiple system atrophy with high sensitivity and specificity. Our first reports of the initial findings, at the 143rd Annual Meeting of the American Neurological Association, Atlanta, October 2018, and the Annual Meeting of the Society for Neuroscience, San Diego, November 2018, were featured on MedScape and MedPage. Additional validation of the initial findings in a separate, independent cohort and many additional control experiments led to a 2021 publication of the data and coverage on the UCLAHealth blog. Using measurements of α-synuclein in serum/plasma EVs originating in neurons or oligodendrocytes, each immunoprecipitated using a different specific marker of the cells of origin, we created a composite biomarker including the total concentration of α-synuclein in the neuronal EVs, the ratio between the α-synuclein concentrations in oligodendroglial and neuronal EVs, and the total concentration of EVs in the sample. The formula for this composite biomarker was created using the results of the Discovery cohort to give the optimal separation between the group. It was then applied to the independent Validation cohort and yielded 89.8% sensitivity and 86.0% specificity for separating PD from MSA, an unprecedented separation power between these two diseases using a blood test.
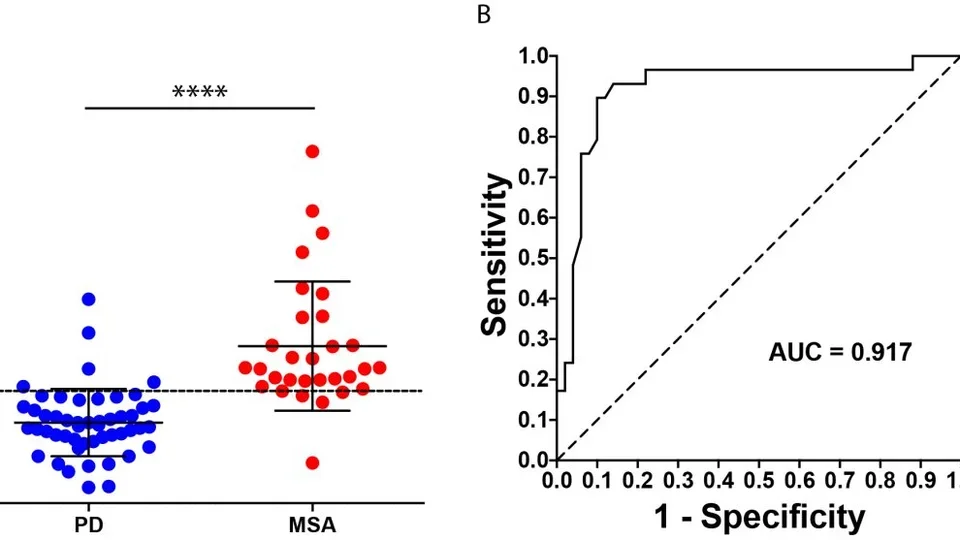
A blood test separates Parkinson's disease (PD) from the related synucleinopathy, multiple system atrophy (MSA) improving the diagnosis of these diseases. For more details, see Dutta et al., Acta Neuropathologica, 2021.
In a follow-up project, we used “leftover” samples from the first study and tested three additional biomarkers in them. Using a new assay we developed for sensitive measurement of a pathological, phosphorylated form of α-synuclein (pS129-α-synuclein), we found that adding the concentration of this biomarker in oligodendroglial EVs to the composite biomarker formula improved the separation between PD and MSA to 89.4% sensitivity and 90.0% specificity (Taha et al., 2023).
In a different study, we tested a similar approach for its ability to measure effect of the molecular tweezer, CLR01, in a mouse model of tauopathy. The mice were treated with CLR01 for 5 weeks and showed significant improvement in their muscle strength and disinhibition-like behavior, as well as in different measures of their brain pathology, compared to mice receiving a placebo. Analysis of the protein tau in neuronal EVs isolated from the blood of the mice showed that the concentration of tau increased in correlation with the treatment dose and the improvement in the phenotype, suggesting that this technique can be used not only for diagnosis but also for measuring treatment effect.